Why the 2025 BSMM Best Practice Recommendations Matter – and Why We Must Act Now
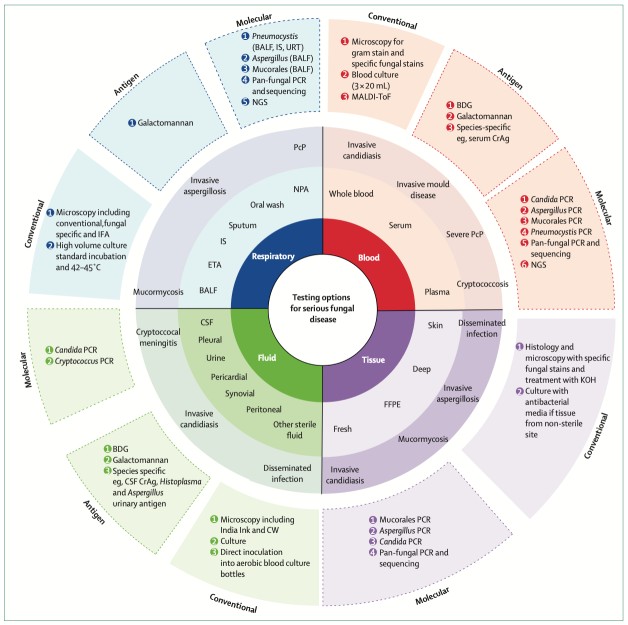
The 2025 British Society for Medical Mycology (BSMM) Best Practice Recommendations for the Diagnosis of Serious Fungal Diseases represent one of the most significant advances in fungal diagnostics guidance in a decade. These standards arrive at a critical moment: fungal diseases are rising, patients are increasingly complex, resistance is growing, and diagnostic delays still cost lives. By integrating cutting-edge molecular testing, faster turnaround requirements, improved mycology workflows, expanded high-risk populations, and a stronger link to antifungal stewardship, these best practices redefine what “good care” should look like in the NHS and in health systems worldwide.
Full paper
This document is more than a guideline—it is a call to action.
Why these guidelines deserve universal adoption
The BSMM recommendations build on the reality that culture and microscopy now diagnose only ~10% of serious fungal diseases. Most cases rely on non-culture diagnostics—PCR, antigen, antibody and BDG assays—whose correct use can drastically improve early recognition, reduce unnecessary antifungals, and save lives. The guidance establishes:
- Expected turnaround times and auditable standards
- Expanded use of galactomannan, BDG, and Pneumocystis PCR
- Clear recommendations for high-volume cultures and mycology-specific specimen handling
- When and how to apply fungal IgG and IgE
- How to combine tests to confirm or exclude disease in real-world clinical pathways
- Integration with antifungal stewardship—essential for preserving limited antifungal classes
- Adaptations for resource-limited settings
The document also emphasises the need for active communication of positive results within 2 hours, a standard that—if adopted universally—has enormous potential to change clinical outcomes.
25SS BSMM Best practise
In short, this is a blueprint for safer, faster, more accurate fungal diagnosis in every healthcare system.
Who needs to hear this—and why
To create real-world change, these recommendations must reach the right people. This includes both clinical decision-makers and system managers:
1. Hospital laboratory directors and pathology network leads
They determine what tests are commissioned, which samples are processed locally, and how fast results are delivered.
→ Message: These standards are auditable, evidence-based, and aligned with ISO15189:2022. Meeting them strengthens your service, your accreditation position, and patient safety.
2. NHS ICBs, regional clinical leads, and commissioners
Fungal diagnostics are often invisible in commissioning discussions.
→ Message: Adopting BSMM standards reduces inappropriate antifungal use, shortens hospital stays, and prevents ICU deaths—directly lowering costs.
3. Respiratory, ICU, haematology/oncology, transplant and infectious disease clinicians
These are the groups who most often face fungal disease and who depend on test accuracy and speed.
→ Message: This guidance clarifies the right tests, the right combinations, and the right patient groups. It will directly improve your diagnostic certainty.
4. Allied health professionals and advanced clinical practitioners
Increasingly involved in early assessment and triage.
→ Message: Understanding when fungal disease should be suspected empowers earlier sampling and avoids missed diagnoses.
5. Global public health agencies and hospitals in low- and middle-income countries
The BSMM group has included a tiered, resource-adaptable pathway.
→ Message: Even with limited resources, these recommendations guide stepwise improvement and provide a defensible roadmap for scaling up diagnostics.
6. Patient organisations, advocacy groups, and rare disease networks
Well-informed patients and carers are powerful drivers of change.
→ Message: These standards help ensure patients receive timely, evidence-based diagnosis, reducing years of misdiagnosis and unnecessary treatment.
What will make the biggest impact
A. National and regional dissemination
- Publish short summaries through NHS England, UKHSA, ICS/ICB bulletins, and RCPath networks.
- Encourage trusts to include the guidance in Quality Improvement and clinical governance programmes.
B. Integrate into antifungal stewardship
Every hospital prescribing antifungals should incorporate these recommendations into its stewardship protocols.
This links diagnostics directly to clinical decision-making and resource use.
C. Education, webinars, and clinical-pathway updates
Deliver focused education sessions for:
- ICU teams (influenza-/COVID-associated aspergillosis)
- Respiratory teams (CPA, ABPA, bronchiectasis)
- Oncology and transplant teams
- Paediatric services
D. Embed in electronic systems
Work with EPIC/HIVE and other EPR systems to:
- Prompt clinicians with the correct test panels
- Flag recommended turnaround times
- Ensure positive results trigger rapid alerts
E. International amplification
- Share through ECMM, ISHAM, IDSA, ESCMID, Global Action for Fungal Infections (GAFFI), and African/Asian mycology networks.
- Encourage adoption as a global reference standard, particularly where fungal disease burden is highest.
F. Patient-facing routes
Patient groups can help push for adoption by:
- Sharing accessible summaries
- Encouraging hospitals to benchmark themselves
- Asking clinicians whether fungal diagnostics meet BSMM standards
Conclusion: Implementing these standards will save lives
The 2025 BSMM recommendations are a landmark achievement. They bring clarity to a complex diagnostic landscape and offer a pragmatic route to improving outcomes across specialties and healthcare systems.
The most powerful next step is ensuring these recommendations leave the page and enter everyday clinical practice—everywhere.
Adoption of these best practices means:
earlier diagnosis, fewer missed cases, more targeted antifungals, improved survival, and better stewardship of precious antifungal resources.
This is a moment for leadership across UK and global health systems.
The tools exist. The evidence exists.
What we need now is implementation.
